[Image credit: Aaswath P. Raman]
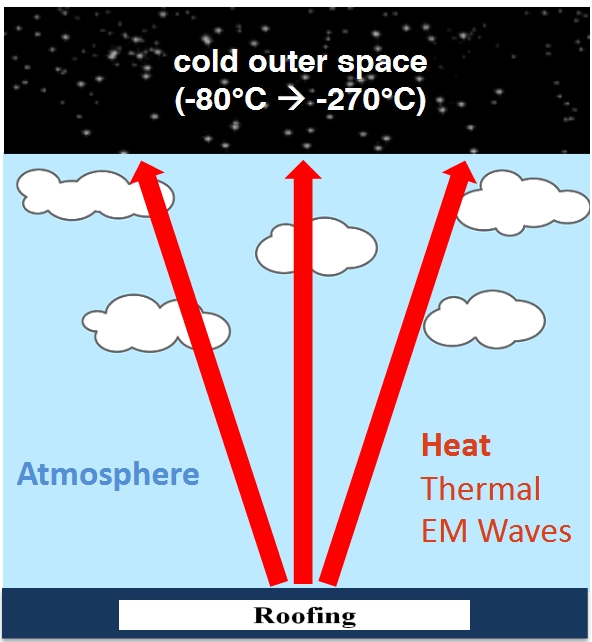
Let’s say you walk into an unheated room. The air temperature is only 50°F. There is a wood stove in the room, but the wood stove is unlit.
Your body is at about 98°F. Since your body is warmer than absolute zero, it is radiating heat in all directions. (All objects that aren’t at absolute zero emit infrared radiation). The wood stove is cool — it’s at 50°F. Still, the wood stove is also radiating heat in all directions.
Since you are warmer than the wood stove, you are radiating more heat to the wood stove than the wood stove is radiating to you. If you stand there long enough, the wood stove will get a little warmer, and you will get a little colder.
If someone lights a fire in the wood stove, and the temperature of the stove rises to 300°F, what happens? You are still radiating heat to the wood stove. And the wood stove is still radiating heat in your direction. But since radiant heat is flowing at a faster rate from the stove to you than from you to the stove, you are going to end up feeling warmer.
The phenomenon I’m describing is explained this way in a document posted on the National Weather Service web site: “Every warm object loses energy by radiating electromagnetic energy (e.g., infrared photons). If it receives an equal amount of energy from other objects, it is in radiative equilibrium; if it receives less from other objects, it loses energy and cools down.”
An online article by Mike Luciuk notes, “The outgoing radiated power of [an average human] would be about 1,039 watts. In other words, a human body radiates power close to that of a toaster, with a peak wavelength of about 9.5 µm.…
Weekly Newsletter
Get building science and energy efficiency advice, plus special offers, in your inbox.

This article is only available to GBA Prime Members
Sign up for a free trial and get instant access to this article as well as GBA’s complete library of premium articles and construction details.
Start Free TrialAlready a member? Log in













29 Comments
Recommendations?
Wondering what building practices would you recommend, such as:
-- Cool roof helps reduce AC costs and reduce summertime overheating
-- For ventilated attics in humid climates, avoid OSB sheathing (more subject to moisture damage) and cellulose insulation (absorbs moisture, may contribute to attic humidity and mold?), ...
-- For unvented roofs, use exterior rigid insulation and ...
Response to Robert Opaluch
Robert,
I'm not sure what problem you are trying to address.
Are you talking about a real building? If so, tell us your climate zone, and tell us more about the configuration of the roofs and the preferred location for the insulation layer.
Timely Article
We are very close to replacing old asphalt shingles with a standing seam metal roof. Could one of the building experts here answer my question. Thanks in advance.
Should insulation be applied underneath a SS metal roof (above sheathing) in a mixed humid climate (S. IN)?
My plan is high temp peel and stick underlayment over 1/2" plywood deck, followed by 24 g SS metal roof attached with hidden brackets. Color choice is copper penny (45% reflectivity), gabled roof has 5/12 pitch and faces E-W. E facing is full sun and west side is heavily shaded. E side will have solar PV panels attached after metal is installed. Full soffit venting along eaves (24" wide vent) and full ridge vent. R70 insulation in sealed home attic, but no insulation in attached, unheated garage that will also get metal roof. Work will be done by licensed roofing contractors.
Any better ideas?
Response to Joe Dwyer (Comment #3)
Joe,
I agree with Malcolm. You have a vented attic, and you don't live in the Pacific Northwest. There is absolutely no reason to believe that your roof sheathing will be damp or moldy.
Building codes require the installation of roofing underlayment (asphalt felt or a synthetic roofing underlayment) between the roof sheathing and the roofing, but there is no need to install peel-and-stick underlayment under standing-seam metal roofing.
Delete
Delete
Martin or Malcolm
Thanks for the quick response.
The idea with using peel and stick underlayment instead of synthetic is two fold. First, the roof will be walked on after roof install (solar, maintenance) and I didn't want any fastener or washer denting or damaging the underside of the metal roof. Second, in the event of worse case storm where the metal is blow off, then peel and stick will more likely stay down and offer water protection, whereas synthetic won't. Peel and stick is only about $300 upcharge to synthetic underlayment, at least for the Corning hi temp. brand designed for underneath metal roofs.
Comments?
How does this apply to RVs?
I had a travel trailer with a white, flat roof that rotted apart even with repairs done to the roof. PacNW, zone 4c, 2 - 3" of fiberglass in the ceiling (maybe).
Can what you described here happen to RVs?
Would keeping the trailer in a carport help?
Response to Joe Dwyer (Comment #6)
Joe,
A peel-and-stick underlayment will work, as long as you choose a product designed to withstand the high temperatures under metal roofing -- for example, something like Grace Ice & Water Shield HT.
Response to Mark Walker (Comment #7)
Mark,
I'm sorry, but I can't diagnose the causes of your roof rot problems based on the limited information you have provided.
If there is sheathing rot under a flat roof, a roof leak would certainly be the first thing to investigate. But it's also possible that condensation played a part.
Good read.
Excellent article! Thanks as always. Building science isn't rocket science, but I'll be darned if it isn't at least as complicated...
Dwyer issue
Martin,
I'm in a similar spot as Mr. Dwyer, wanting to install a standing seam metal roof (somewhat similar climate-northern part of zone 4 in VA). Like him I was pondering whether a peel and stick membrane may offer extra insurance against moisture while reading this article. One major difference, I'm considering an unvented roof, possibly with foam on the underside of the sheathing. I suppose closed cell foam would be a no-no with Grace Ice & Water Shield. But it seems, Martin, that you are making the case against an air space between the metal roofing and sheathing regardless. Is that correct? Because I was initially thinking an air space between roofing material and sheathing was a good idea. Now I'm somewhat puzzled.
Response to Antonio Oliver
Antonio,
I'm not sure why you concluded that I am "making the case against an air space between the metal roofing and sheathing." Such an air space is usually a good idea.
If your roof assembly has such an air space, you can expect condensation on the underside of the metal roofing under some weather conditions. That's why our building codes require, and building scientists recommend, that you have a layer of roofing underlayment between the roof sheathing and the air space.
The condensation drips on the underlayment, and usually evaporates before it causes any problems.
Closed cell foam under the roof deck @ Antonio Oliver # 11
"I suppose closed cell foam would be a no-no with Grace Ice & Water Shield."
That's a common misconception.
As a general rule most roof assemblies don't dry toward the exterior. A typical #30 felt + asphalt shingle layup runs about 0.1 perms, on the verge of being a true vapor barrier. Taking it down to 0.05 perms or less doesn't make much of a difference.
The low vapor permeance of closed cell foam applied to the under side of the roof deck inhibits drying toward the interior, but it also protects the roof deck from moisture drives from the interior that occur whenever the roof deck's temperature is below the dew point of the interior conditioned space air. As long as the closed cell foam is applied when the roof deck's moisture content is low enough, it's not a problem.
Closed cell foam is one of the least-green insulation products out there, primarily due to the industry-standard HFC245fa blowing agent use (there are a few exceptions), but also because of the high polymer per R ratio. As such it's greener to use the minimum amount that ensures a moisture safe stackup.
In zone 4A only ~30% of the total R needs to be low permeance foam on the exterior to limit the risk of moisture build up in higher permeance insulation materials applied directly to the underside of the foam, and that is built into the IRC prescriptives for unvented roof. The IRC requires a minimum of R15 out of a code minimum R49 (which 30.6% of the total) to be low permeance on the exterior of fiber insulation or open cell foam. So you can get to a code minimum R49 with 3" of closed cell polyurethane (R18-R21) and R28-R30 of fiber directly below and in contact with the closed cell foam. That's definitely greener, and usually cheaper than doing it with 7-8" of closed cell foam too.
At 3" most closed cell foam will run about 0.3-0.4 perms, which is sufficiently vapor open in a zone 4A climate that any seasonal accumulations of moisture can dry in a reasonable amount of time toward the interior, but sufficiently vapor tight that the peak moisture levels in the sheathing over a winter are well controlled to a safe level.
HFC blown closed cell foam can only be safely installed in 2" lifts, with a cooling period between lifts for fire safety as it cures. Though some contractors will take the fire risk and try to cheat that, the higher temperatures during cure becomes a quality issue as well. The newer much lower impact HFO blown foams can be blown in depths of 4" or more, and tend to be a bit higher-R (~R7/inch). Even though it's usually a bit more expensive to go with HFO blown foam, for a 3-4" installation it might come in about the same, since it can be done in one pass, not two, and thus no cooling period is required.
Thanks Dana and Martin
As usual, you guys have advice backed up by solid reasoning.
Martin, I suppose I thought you were recommending avoiding the air space between metal roofing and the roof deck because it's that air space that allows moist air to get between the roofing and the deck in the first place. Thus, I reasoned that eliminating that air space eliminates (or at least greatly reduces) the chance of of water condensing onto the underside of the metal roofing. Does that make sense? BTW, I see a lot of metal roofing installed in my area with no spacing between the roofing and the roof deck. But thanks for the clarification.
And, Dana, it is not intuitive to think that sandwiching plywood between metal and rubber/metal foil would not be a bad idea. Thanks for your explanation.
Here measured some drying to
Here measured some drying to the exterior advantage to permeable underlayment.
"...the outer moisture content in the vapour impermeable assembly
is consistently wetter than the permeable assembly" (figure 20).
My guess is that ventilated Pacific NW attics would benefit from smart, pressure balanced ventilation. Ie, stop moving air through the attic under condensing conditions. But maybe simply painting the sheathing underside (Table 4) is more cost effective.
Jon
My own experience with mouldy sheathing here in the PNW is that in ventilated attics with good air-sealing between it and the conditioned space below, although you do get some surface discolouration, it isn't a concern. You see about the same level on the underside of un-heated outbuildings, or structures, like carports, open to the elements. Where the problem becomes acute is typically confined to poorly air-sealed attics and cathedral roofs. All of which is a long way of saying the study seems to get it right.
Two things that seem to influence the mold growth are the orientation of the roofs and the micro-climate of the site. Shady forested lots understandably see more.
Traditionally most roofs here were either metal on purlins, or shakes installed on skip sheathing. Both strategies seem to eliminate the problem entirely. The other thing that proved remarkably effective was the absence of any air-sealing at all. Very old structures, like our local community hall, leak such vast amounts of warm air in all directions that they experience no damage. Of course the energy penalty is tremendous.
Since our code here mandates the use of panel goods for roof sheathing, that leaves us with the unpalatable option of applying a coating to the underside of the sheathing - or perhaps a manufacturer will begin marketing a product with a factory applied barrier. Until then simple roofs with adequate ventilation seem to be the best option.
There is LP TechShield (foil
There is LP TechShield (foil covered OSB), although it may not be available there.
Jon
Thanks. Somethng like that might make the difference, although it is starting from a more vulnerable point using OSB as its base than plywood.
Blue-Green people?
Martin, I can’t help myself.
You state the following above: “In other words, a human body radiates power close to that of a toaster, with a peak wavelength of about 0.5 µm.” But the peak in radiated power from the human body occurs at a wavelength of about 10 µm. (the article you site states 9.5 µm) The peak in the emission of a toaster element is about 2 µm. It’s the sun’s radiation that peaks at 0.5 µm (or so). Here’s a pretty good plot of the radiated power vs, wavelength for the sun, a toaster element (ish) and a warm room (or a cold person):
http://quantummechanics.ucsd.edu/ph130a/130_notes/node48.html
Response to Bill Dietze
Bill,
I am indebted to your sharp eyes and knowledge of wavelengths. Thank very much for catching the typo; I have corrected it.
3 Celsius not 3 Kelvin
Another nitpick.
The radiative night sky temperature is (by your reference) 3.9 Celsius, not 3 Kelvin. The microwave background radiation of the universe is about 3 Kelvin, but our atmosphere is much warmer than that. The roof is thus in radiative exchange with the earth's night sky, not with deep space. Otherwise we'd be losing heat a lot faster.
Response to Kathryn Oseen-Senda
Kathryn,
I'm grateful for your correction. The error was due to my misunderstanding; I have corrected it. Thanks.
Night Sky Radiation
Harold R. Hay, inventor of the original "Sky Therm" roof pond cooling system and of the process for pressure treating wood once commented that British officers stationed in India would place trays of water on the north sides of buildings overnight so they could have ice for their gin and tonics.
We are currently monitoring the undersides of California roof decks in "high performance" vented attics that have fiberglass insulation applied to both the underside of the deck and the ceiling to see if the dew point temperature is approached or if condensation occurs.
Response to Frontier Energy
If British officers in India were making ice outdoors at night, they must have been stationed at a high elevation in the Himalayas. The air temperature has to be close to freezing to make ice that way.
Either high, or very dry
With very clear dry air with a dew point below freezing the air temp can be quite a bit above freezing and still be able to create ice at night. I've seen overnight skim-icing and some frost at only moderate elevation while camping in AZ, when it had hit 90F the prior afternoon, but that may not be very common. I'm not sure if the not-so-elevated deserts of Rajasthan or Gujarat are quite that dry on a regular basis either, but they're pretty dessicated. The gin & tonic ice tray story sounds a bit apocryphal, but may have grown from overnight skim layer icing observations such as that.
"frigorific rays" is my new
"frigorific rays" is my new favorite phrase.
Response to Leigha Dickens
Leigha,
I've spent many nights outdoors when camping without a tent, and I've shivered in my sleeping bag, suffering the effects of those darned frigorific rays.
Leigha
If I decide to change careers and become a blues musician I think I'll call myself Figorific Ray.
Great article. I know this is old but... science.
"a warm object on earth can get cold fast — colder, in fact, than the temperature of the outdoor air. Why? Because outer space is cold. The sky has an effective temperature of only 3.9°C."
This appears to be a corrected statement made in response to Kathryn Oseen-Senda, and is pulled from the linked article: https://asterism.org/wp-content/uploads/2019/03/tut37-Radiative-Cooling.pdf
The 3.9°C figure appears to be from a specific calculation based on specific conditions (namely a cloudless 10°C night with 25% humidity). Unless I am misunderstanding, it is NOT some 'radiative sky constant' temperature.
"The roof is thus in radiative exchange with the earth's night sky, not with deep space. Otherwise we'd be losing heat a lot faster." - Kathryn
Sort of, but this feels confused. We are ALWAYS radiating—to what we are radiating is irrelevant. What is relevant is what is radiating back to us. Deep space certainly is not (infinitesimal), though the atmosphere is.
The introductory story was spot on. As a human body, you emit radiation towards the stove and the stove emits towards you; whether you are cooled or warmed depends on who is emitting more (the stove or you), but in simplistic terms, the only variable here is the stove. That's why the clearest nights show 'increased radiative cooling' because there is less being radiated back vs when there are clouds. The roof isn't radiating 'more,' its just getting back less. Space being really cold matters insofar that it isn't offering radiation back, until we spin far enough to face the sun.
A related point is that the reason an object can become colder than ambient has to do with the emissivity constant, not the fact that space (or the atmosphere) is very cold. Otherwise, wouldn't the air (ambient) also radiate to the same very cold space and therefore match the temp of the roof? A true 'black body' has an emissivity constant of 1, human body about 0.95, and the night sky 0.74. I was struggling to understand—and finding no satisfactory answers I could comprehend—how an object could actually become colder than its surrounding due simply to distant space being cold. I thought, 'how can it know or care how cold space is?' It doesn't, it just gives at a higher rate than the air around it, and space has practically nothing to give back to the roof or the air.
Log in or become a member to post a comment.
Sign up Log in