
The 22nd annual Westford Symposium on Building Science (a.k.a. Building Science Summer Camp) happened two weeks ago. And it began with a bang, as Professor John Straube expounded on moisture physics for half a day. I’ve now had the chance to hear him present on mechanical systems for low-load homes (at the 2012 BSC Experts’ Session), air barriers (at the 2016 Air Barrier Association of America conference), and this time on moisture fundamentals. I’m sure there is no one in the world better at explaining the fundamentals of building science, making it relevant to what we do with buildings, and inserting a healthy dose of Straubisms to make you laugh while you learn from the master.
If you’ve ever heard Professor Straube speak, you know he can pack a lot into a short time. Since he spent half a day on moisture physics, there’s no way I can give you thorough report on it in one little blog article. I’ll do what I can to cover the parts I found most interesting and informative. Then you can go download the slides from his presentation to see what else he covered.
Here’s the outline he presented:
- A review of moisture fundamentals
- Focus on materials, not systems
- Details of water/moisture in all its forms
- Interaction of moisture with materials
- Porous materials
- Deeper dives… Wood, concrete, etc.
Ready? Let’s go.
Moisture balance and water fundamentals
The first major concept he covered was the moisture balance. The diagram below shows the three basic processes of wetting, drying, and storage.
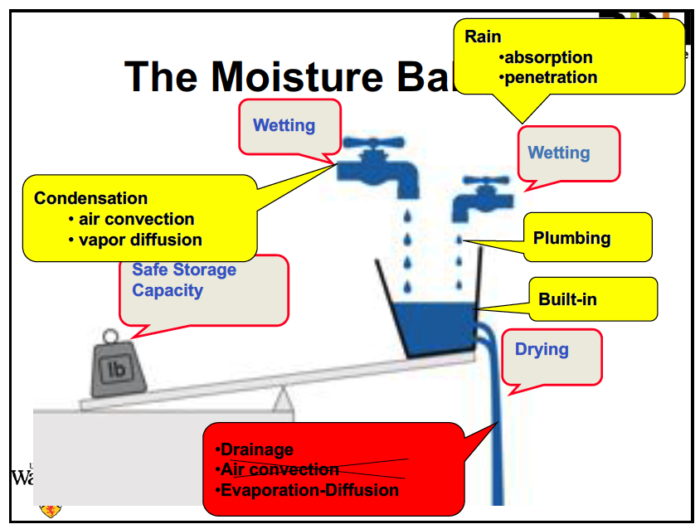
The two types of wetting are by water vapor condensing or adsorbing and by liquid water getting in. The yellow and red boxes show the sources or mechanisms for wetting and drying, but I don’t recall why he crossed out air convection as a drying mechanism and apparently didn’t take notes on that part either. Air convection is certainly a drying mechanism, depending on what the relative moisture levels are, and he discussed how that happens with both intentional and unintentional air convection.
From there he went to the polarity of the water molecule and how that affects the various phases of water. That, of course, led to psychrometrics and phase changes and the fourth state of water: adsorbed water.
And here he shed some light on what many find confusing when they first learn about adsorption. The difference between adsorbed water and absorbed water, Straube said, lies in where the water came from. When water vapor attaches itself to the surfaces of porous materials, that’s adsorption. When liquid water gets into the pores of a material, that’s absorption.
One of the first things Straube talked about was the importance of using the right words. Another confusing term is condensation, because that’s really the process that happens on nonporous materials like glass or metal. When water vapor attaches itself to the surfaces in porous materials, that’s adsorption. Professor Straube provides an out here, though, since everyone uses the word “condensation” to describe adsorption. Capillary condensation, he said, is what happens when water vapor adsorbs to porous materials.
Speaking of capillary condensation, Straube said that Houston in summer is worse for such problems than Vermont in winter. That doesn’t mean capillary condensation in Vermont is OK. Because of the lower temperatures, Vermont just won’t be as likely to support the microbial infestations that rot wood and cause indoor air quality problems. But when capillary condensation occurs on the backside of air conditioned drywall in Houston, the temperature is much more conducive to microbial growth.
Water clusters
One interesting property of liquid water you may not be aware of is that it’s not just a continuous mass of water molecules. The water molecules group themselves into large clusters. Because water molecules are polar, having one end with a net positive charge and another end with a net negative charge, they attract each other, positive to negative and vice versa. In the liquid phase, the molecules constantly realign themselves because of thermal motion. But even with the constant shifting of allegiances, molecules cluster into groups, with the size of the clusters related to temperature.

This is not only interesting by itself, it also has application in building science. In a material like spun-bonded polyolefin, the stuff that Tyvek house wrap is made of, liquid water has trouble going through the pores because of the size of the water clusters.
Material wetness
Straube spent quite a bit of time discussing material wetness. He began by introducing adsorption isotherms (often called sorption isotherms), which plot the moisture content of a porous material as a function of the relative humidity around the material. This is all done at constant temperature, hence the “isotherm” part of the name.
First, percent moisture content (MC) is relative to dry weight of the material. That’s why concrete is ~5% MC at ~95% RH, which is equivalent to the moisture content in wood when it’s at ~17% MC.
So now take a look at the sorption isotherm below. He didn’t say what the material is but sorption isotherms mostly have that same general shape. They also have hysteresis, meaning the moisture content is different as you dry it out than it was as you were wetting it.
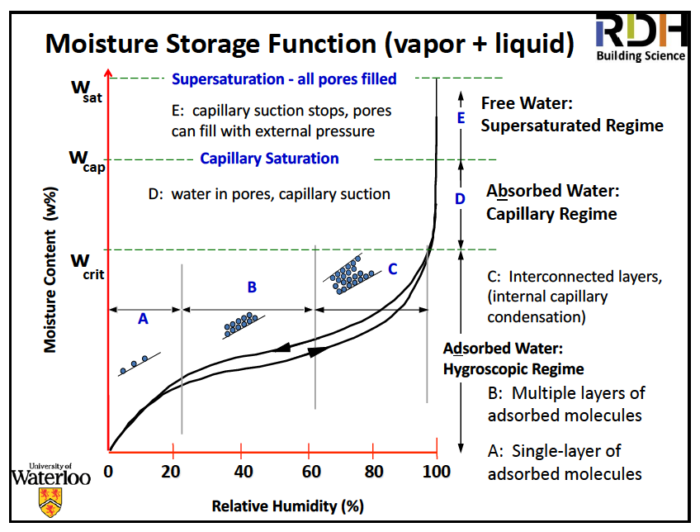
The other thing to notice on that diagram is the three moisture regions:
- Hygroscopic – dry to the touch
- Capillary – wet to the touch but doesn’t drain
- Supersaturated – wet to the touch; does drain
These regions are significant because of the different things that happen in them.
- Mold and corrosion — Tend to begin around 80% RH when the water layer is thick enough that “free” H20 is available
- Swelling and shrinkage — only occurs in hygroscopic range
- Freeze/thaw — capillary range
Moisture in wood
Professor Straube brought all this fundamental moisture physics home with a discussion of concrete and wood, two materials used occasionally in buildings. (And when I say “occasionally,” I mean in almost every building.) He talked about porosity and permeability and how those properties affect the ability of a material get wet, store water, change size, deform, and dry out.
Concrete is kind of boring because it’s generally an isotropic material. Wood on the other hand is anisotropic. Isotropic materials are the same no matter which direction you look. Anisotropic behave differently when you measure a property in different directions.
Wood is like the Internet, a series of tubes. Water moves easily in the direction of the tubes, not so easily across the tubes. That’s why wood is a good material to make boats and casks out of. Shrinkage of wood begins when its moisture content drops below about 28%. Once it dries out, it can swell again through adsorption or absorption of more water.
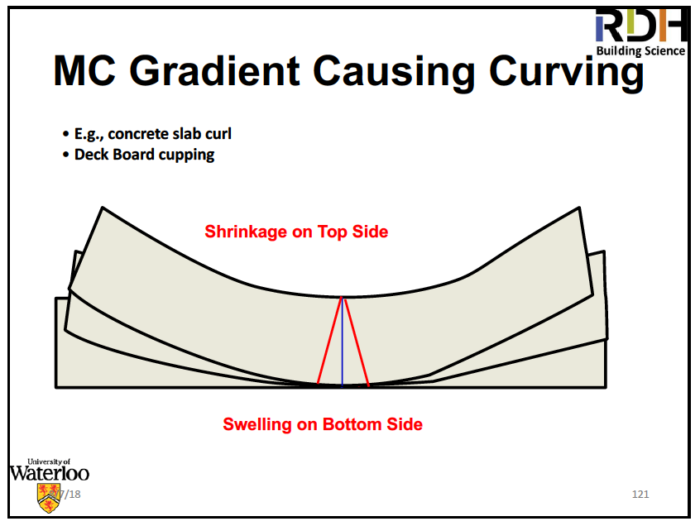
Shrinking and swelling of wood as it dries out and gets wet can be a problem if you don’t manage it properly. Straube’s advice in this area is the following:
1. Allow movement
2. For cupping:
- Reduce moisture content gradient across wood
- Reduce thickness
- Reduce stiffness and restrain
3. For shrinkage and swelling:
- Reduce range of relative humidity
- Reduce rate of change
- Reduce cross-grain!
As Dr. Joe Lstiburek says, “Wood is good… but strange.”
Some of the best Straubisms
Of course, no talk by Dr. Straube is complete without a healthy dose of Straubisms sprinkled throughout the talk. Here are the ones I managed to record:
“If you meant elephant and you said rhinoceros, it’s not the same thing.” (Part of his opening lesson on the importance of language.)
“Nails make about one hole per nail.” (Because sometimes they make more than one hole.)
“In the old days… the good old days when people died at 40 with no teeth…”
“[Before air conditioning in Houston], the technique they used for human comfort is a concept we call suffering.”
“Like Joe says, even the dumbest of the three little pigs didn’t build his house out of paper. Then all the wolf has to do is come over and lift his leg.”
“Our children are precious so we must create as much mold as possible [in schools].”
He’s quite the entertainer.
A deep well
The material Professor Straube covered in this half-day presentation could easily be expanded into a semester-long course on moisture physics. He covered much more than I’ve written about here, as you can tell if you download the slides. There’s a heck of a lot there and if you want to go further without taking such a course, a good alternative would be to study the book Water In Buildings by Bill Rose.
After three and half hours on that Monday morning in early August, Straube concluded thus:
“Moisture is worth understanding.”
“Porous materials are common… and much more porous than you think.”
I concur.
Allison Bailes of Decatur, Georgia, is a speaker, writer, building science consultant, and the author of the Energy Vanguard Blog. You can follow him on Twitter at @EnergyVanguard.
Weekly Newsletter
Get building science and energy efficiency advice, plus special offers, in your inbox.














5 Comments
John Straube has recorded 9 hours of video presentations covering fundamental building science principles that are available through the PG&E Energy Training Centers. These on-demand trainings promote durability, comfort, energy-efficiency, and reduction of greenhouse gas emissions buildings. John includes some discussion of energy standards applicable to design and construction in California. PG&E is the electric/gas utility providing service to much of Northern California. These trainings were funded by California utility customers and administered by PG&E under the auspices of the California Public Utilities Commission.
You must first create a log-in with the PG&E Energy Training Centers and can then register for any class in this series. Go to http://www.pge.com/energyclasses and select the 'button' for on-demand trainings near the top of that page to register. Below are the titles of the sessions in this series with links to a registration page for each individual session.
Building Science 1.0 Overview and Introduction to Control Layers - 2 hours
http://usi.pge.com/event-details?EventID=17920
Building Science 2.1 Introduction to Heat Transfer - 1 hour
http://usi.pge.com/event-details?EventID=19142
Building Science 2.2 Airtightness and Air Barriers - 1 hour
http://usi.pge.com/event-details?EventID=19143
Building Science 2.3 Understanding and Limiting Thermal Bridging - 1 hour
http://usi.pge.com/event-details?EventID=19144
Building Science 2.4 Introduction to Continuous Insulation and Cladding Attachment - 1 hour
http://usi.pge.com/event-details?EventID=19145
Building Science 2.5 Introduction to Windows, Curtain Walls, Window Walls and Shading Design - 1 hour
http://usi.pge.com/event-details?EventID=19146
Building Science 2.6 Introduction to Moisture and Buildings - 1 hour
http://usi.pge.com/event-details?EventID=19147
Building Science 2.7 Understanding the Psychrometrics of Condensation - 1 hour
http://usi.pge.com/event-details?EventID=19148
Building Science 2.8 Introduction to the Control of Rain and Groundwater Penetration - 1 hour
http://usi.pge.com/event-details?EventID=19149
Full disclosure, I am an architect and work at the PG&E Energy Training Centers. I engaged John to record these sessions.
Bill Burke
Bill,
Thanks for the links. GBA readers can also check out several GBA podcasts by John Straube, including these:
"Why Does Green Building Matter?"
"Why Does Green Building Matter? (Part 2)"
"How Air Affects a House (1)"
"How Air Affects a House (2)"
"How Heat Moves Through Homes"
GBA also has a podcast that features an interview with John Straube. Here is the link:
"The Green Architects Chat With John Straube"
This gives quite a lot for us to think about when it comes to the materials that surround our crawl spaces and are used for the ducting in our home. Especially for those of us who live in colder climates where we are advised to use devices such as furnace humidifiers to offset the dry air caused by using our furnaces such as shown: https://heattalk.com/furnace-humidifiers/
This article highlights that we should be aware of whether the materials in vital parts of our home are hygroscopic, capillary or supersaturated when we use these devices as we could be doing more harm than good in an attempt at alleviating or preventing respiratory problems.
Even in cold/very-cold climates in a tight house with a well balanced duct system a humidifier isn't usually needed to keep the indoor RH above 30% unless the ventilation rate requirement is quite high.
Unbalanced duct systems drive excessive outdoor air infiltration while operating, and humidifiers are often installed as the bandaid solution to to a mediocre duct design/implementation, when the better solution would be to correct the deficiencies of the duct system or air-leakage of the building envelope. An Energy Star duct system requires that the room to room pressure differences not exceed 3 pascals (0.012" water column) under all operating conditions, room doors open/closed, etc. Most systems not designed per Manual-D will have rooms hitting several times that much. When that happens a significant part of the return path is via "the great outdoors", unless the house itself is practically hermetically sealed tight.
I am hoping to build a house using hempcrete and there is presently much promotion of the neccesity for breathable envelopes in buildings but with little real information. How would I go about knowing whether this material is hygroscopic or capillaric and how would this affect internal humidity in the building especially during the colder months, also the rainiest months here in Scotland, when using air to air heat pumps?
Log in or create an account to post a comment.
Sign up Log in