Airtightness and Vapor Permeability
Can anyone point me to an answer that explains the physics? Obviously to be vapor permeable a material must have penetrations that are bigger than vapor molecules; why doesn’t air, driven by an air pressure differential, flow through the same holes? I’ve heard it said that the pores are big enough for H2O molecules but too small for O2; is that it? Or is it simply that the rate of air leakage is too low to matter given the grosser methods used to measure air leakage compared to vapor diffusion? Or is there some force binding the molecules in a volume of air that prevents them from convecting through very small openings. Or perhaps a force greater than air pressure that retards them within the pores? I discovered in Straube’s Building Science for Building Enclosures that liquid water tends to exist in clumps of about 80 molecules and that this phenomena allows for materials to be at once watertight and vapor permeable. Is there some similar clumping phenomena occurring in air?
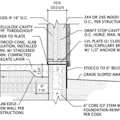
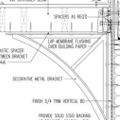
GBA Detail Library
A collection of one thousand construction details organized by climate and house part







Replies
Hayden, check out comment #16 & 19 by Riversong at this blog
https://www.greenbuildingadvisor.com/community/forum/energy-efficiency-and-durability/15776/vapor-flow-light-frame-construction?page=1
There are also some relevant videos at KhanAcademy...
I will post a link later for the Khan explanation
Hayden,
I'll defer to any chemists who want to chime in. But I think it's easy to have an intuitive understanding of the difference between an air barrier and a vapor barrier by considering a piece of gypsum drywall, which is clearly an air barrier but is also vapor permeable.
We've all seen what happens to drywall if you put it in a hot, steamy environment: it gets damp. In fact, the dampness pervades the material -- it just takes on moisture. If you have a steam room with drywall on the walls, and no other vapor barriers or insulation, clearly the backside of the drywall will evaporate moisture. So drywall is an air barrier but not a vapor barrier.
Hi Hayden,
I believe that certain amounts of air can move through membranes like goretex, tyvek, etc, but as the post above mentions, there isn't a large enough driving force for it to be really noticeable.
The materials are 'breathable', because water vapor can be driven through by vapor pressure differential, which can be quite strong.
Bulk water is kept out because there is a lack of driving force, and because surface tension keeps the capillary tendrils from going too far, if the holes are small enough. I think that you could drive liquid water through these materials if you set up enough pressure differential.
It makes sense to me now: typical vapor pressure differences are much bigger than typical air pressure differences.
Also, I wasn't aware of KhanAcademy and was glad to find that resource.
Thanks everyone!
Hayden
Hi Hayden, great question! I had the same question. Can you post a link to the Khan Academy video? I can't seem to find the right one easily.
A pound of air at 75F and 100% humidity contains 132 grains of air. A grain is a unit of measure equal to 1/7000 of a pound, so that air contains 1.3% water. The amount of water vapor that needs to flow to go from very humid to very dry is miniscule compared to the overall quantity of air. A material that blocks 99% of air flow would be considered a pretty good air barrier. However, if within that flow water vapor is allowed to move preferentially -- because water molecules are smaller than oxygen and nitrogen molecules, which make up most of air -- then there can also be enough vapor flow for the material to be considered vapor open.