How long will closed-cell polyurethane foam retain its greater-than-5 R-value?
The laws of physics and thermodynamics state that a stable R-value higher than about 5 is impossible to achieve when using air as the trapped gas in an insulating material due to the thermal conductivity of air itself. Most materials using air will be lower than than due to the thermal bridging of the structure itself, which is why EPS has an R-value that tops out in the mid 4s. XPS, polyiso and polyurethane boast higher R-values than this because the trapped gas is not air, but rather, some blowing agent with a lower thermal conductivity than air. There have been various discussions centered around the gas in polyiso and XPS leaking out over time, with the claim that after 50 years XPS will be near the R-value of EPS, and Polyiso will settle down to about R-5.5.
It would be interesting to know how these numbers were calculated or observed, if polyurethane has the same characteristics, and what its asymptotic final R-value is. If one can expect the gas to leak out of a very high-R polyurethane form over some period of time and fall to the level of EPS, that would represent a very severe loss of performance and one’s investment. If one buys, say, a polyurethane SIP with 7″ of polyurethane rated to R-50 based on an R-value of 7.2 @ 75f, having that drop to EPS levels of 4.2/inch over, say, 100 years would represent the loss of 41% of the R-value, all the way down to R-29!
I guess the real question is, “how closed-cell is closed cell?” Should we trust R-values higher than 4 or so to hold up over time and be willing to pay their premium?
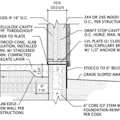
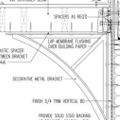
GBA Detail Library
A collection of one thousand construction details organized by climate and house part







Replies
Careful with the "Laws of Physics"!! From the Wikipedia page on Aerogels:
"Aerogels may have a thermal conductivity smaller than the gas they contain. This is caused by the Knudsen effect. Knudsen effect is the reduction of thermal conductivity in gases when the size of the cavity encompassing the gas becomes comparable to the mean free path. Effectively, the cavity restricts the movement of the gas particles, decreasing the thermal conductivity in addition to eliminating convection. For example, thermal conductivity of air is about 25 mW/m·K at STP and in a large container, but decreases to about 5 mW/m·K in a pore 30 nanometers in diameter."
That said, I'd like to know the answer to your questions as well...
Nate,
The short answer is that the problem of thermal drift in spray polyurethane foam exists and has been studied -- see, for examples, this link: https://books.google.com/books?id=AdE8wpSSSKoC&pg=PA17&dq=%22thermal+drift%22+%2B%22spray+polyurethane+foam%22&hl=en&sa=X&ved=0CDEQ6AEwAGoVChMIsrK7gsTpxwIV1RiSCh2UEgFQ#v=onepage&q=%22thermal%20drift%22%20%2B%22spray%20polyurethane%20foam%22&f=false -- but that the problem is generally considered less significant for spray polyurethane foam than for polyisocyanurate.
The answer to the question is complicated by the fact that different blowing agents result in foams with different rates of thermal drift; that blowing agents have been changed by manufacturers over the years; and that blowing agents are likely to change again soon (in response to concerns over the global warming potential of spray foam blowing agents).
As John Straube noted in my recent interview with him (Cold-Weather Performance of Polyisocyanurate), large molecules like Freon stay put longer than small molecules like pentane.