Vapor Retarder vs. Air Barrier
Hi folks,
Would anyone able to clarify the question of Vapor Barrier Vs. Air Barrier?
My struggle is that vapor is a gas, air is a (bunch of) gases. So why wouldn’t vapor barrier imply air barrier?
This one (https://fortifiberflooring.com/product/aquabar-b/) is a class 2 vapor barrier and does not mention any air barrier property. Simple omission because obvious or does air go through? Aquabar-b is two layers of kraft paper laminated with asphalt.
This one https://foursevenfive.com/adhero/ mention 2 separate characteristics:
- Air permeance: 0.00004 cfm/ft2 per ASTM E2178
- Vapor permeance ASTM E96: 8 perms wet cup
Thoughts?
GBA Detail Library
A collection of one thousand construction details organized by climate and house part
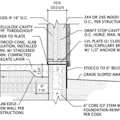
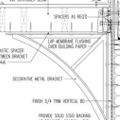







Replies
I think the distinction is that an air barrier is not necessarily a vapour barrier (i.e. tyvek), while a vapour barrier is going to pretty much be both air and vapour barrier (like you say). I'm an amateur though, so keep that in mind.
Thank you for your answer. This describes my intuition as well.
Hard to explain in a couple of paragraphs but an air barrier stops bulk movement of the air (convection) while the vapor barrier stops random individual movements of a given molecule (diffusion).
The bulk movement of a gas happens because there is more pressure at a point. Something pushes the air through your wall, wind for instance. In this case, the entire fluid (air) moves as a whole not only some individual molecules.
On the other hand, diffusion happens without any force. It just the regular random motions of the gas molecules. It occurs because molecules like to spread out from areas of high concentration to areas of lower concentration. Note that here the movement is at the molecule level and not the entire fluid. Only a couple molecules are moving in the same direction not the entire gas.
Convection is a much more effective transport mechanism than diffusion. If there is water vapor in the air, it can move alot of it in a given time period so that's why air barrier is a more important concern than vapor barrier.
So how can a material be an air barrier and not a vapor barrier? I'm no chemist but from what I understand is that the holes in a vapor open membrane are so small that it will only allow a couple molecules to pass which is enough resistance to stop most of the bulk movement of air (and liquid water) while still allowing diffusion to happen.
See
https://www.buildingscience.com/documents/insights/bsi-066-holes-and-leaks
https://en.m.wikipedia.org/wiki/Diffusion
Thank for your answer AlexPoi.
I believe this distinction between Bulk movement and individual molecules is key to understanding.
In my application, I am attempting to block VOC originating from off-gasing of building materials in the floor.
My understanding at this point is that VOCs carried through air currents should be blocked by both products and likely both product will allow for diffusion of those same VOCs, although at a slower rate, as they travel through the barrier.
It seems
My guess is that a material that is a stand-alone vapor barrier is also an air barrier. But it may not be intended/tested/listed as an air barrier.
I'd also say that unless there is no pressure or something else to block air, non-air barriers aren't effective vapor retarders. An interesting practical case is drywall. People paint it and call it a vapor retarder. But if any assembly leaks much air, I suggest that the assembly doesn't serve as a vapor retarder.
Molecular diffusion through a "vapor retarder" is proportional to area. As has been noted, a vapor retarder layer that is 5% holes still is 95% effective at retarding molecular diffusion. It's just that with all those holes, movement of moisture via convective flow (air leaks) overwhelms that via diffusion. It's like the BSC piece that says when it comes to water, don't worry about movement by air leaks until you have kept out liquid water leaks, and don't worry about water movement by diffusion until you have nearly eliminated air leaks.
> a vapor retarder layer that is 5% holes still is 95% effective at retarding molecular diffusion
Exactly. Unfortunately, some people leave off the last 4 words and come up with "A vapor barrier that is riddled with holes that make up 5% of the vapor barrier’s area is still 95% effective".