Water and Wood at Micro and Nano Scale
here is a micrograph of white oak
http://www.flickr.com/photos/54296709@N03/5026397314/in/photostream/
I am trying to verify the scale
Are the units in Microns?
does the visual scale in the lower left corner indicate 100 microns?
Or in other words 100,000 Nanometers?
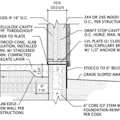
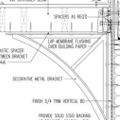
GBA Detail Library
A collection of one thousand construction details organized by climate and house part







Replies
Yes.
http://www.dustboss.com/_data/media/support/how-big-is-a-micron.jpg
Robert,
this illustration and caption from the book Residential Energy
does not seem "right"
http://www.flickr.com/photos/54296709@N03/5025825829/in/photostream/
What do you think?
How would you describe the difference between air migration and vapor migration?
Aren't many of the molecules in the air smaller than water molecules?
Isn't it the high energy of the water vapor (not the size) that gives it the advantage?
The ability to penetrate and or pass thru a network of voids?
I have also been searching for micro-photographs of Gypsum
so far no luck
My John Straube textbook says that a water molecule is about 0.28 nanometers
This 3d-view of birch is interesting
http://www.flickr.com/photos/54296709@N03/5025782421/in/photostream/
these photos are still far,far from nanoscale or water molecule scale
John,
I don't know where Straub got that number, but the bond length between H & O is about 96 pm with the two hydrogen bonds about 105° apart forming a triangle, which makes it smaller than O2 at 121 pm and N2 at 110 pm. A water molecule has an atomic weight of 10, compared to 16 and 14 of O2 and N2, respectively, so it's a bit less sluggish.
http://www.chemistryland.com/CHM130W/12-Gases/AirAtomsMolecules.jpg
But, while oxygen and nitrogen diffuse by kinetic energy alone, water moves both by kinetic energy and surface tension from the positively-charged hydrogen that bonds to other charged substances and surfaces. In liquid form, the incredibly strong surface tension and cohesion of the water creates a strong capillary tension that can move water to great heights against gravity.
But what really causes water vapor diffusion is differential vapor pressure. All air contains about the same relative volume of the various gaseous constituents, but highly variable amounts of water vapor. So there is no partial pressure differential for the other gases, but potentially high partial pressure differential for water vapor.
Wind pressure and stack effect pressures will drive air molecules through "solid" substances, but those are typically in the blower door range of 50 pascals. With interior 68° and 40% RH and outdoor 0° and 90% RH, the vapor pressure differential is nearly 800 pascals.
If anyone else wants to get really small(or really big)...here you go
http://htwins.net/scale2/
I am still searching for a good Micrograph of Gypsum (with a scale indicator)
John,
Thanks for the fun link.
Unfortunately I can't help you out with the micrograph you're looking for.
Take your pick: http://tinyurl.com/gypsum-micrograph