Capturing minisplit condensate in sink in same room
Hello All,
Currently the condensate from the mini-split in the main level of my home is discharged into the kitchen sink for washing dishes etc. Wondering how much of it would typically get evaporated back into the drier air in the room being cooled by the mini-split.
If it’s the case that most of it would get evaporated back into the air, I will let the condensate simply flow down the sink drain.
Thanks in advance.
GBA Detail Library
A collection of one thousand construction details organized by climate and house part
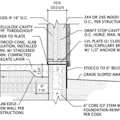
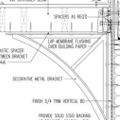







Replies
Hi Venkat.
I'm not sure what your question is. Please clarify...
Hi Brian,
We plug the kitchen sink drain using the stopper and we have the condensate from the mini-split wall unit (which is close to the kitchen area) discharge into the sink. We then use that condensate water to clean the dishes (before the dishes/utensils are loaded into the dishwasher for final soaping/rinsing).
I understand that if there's an open vessel with water in it, that the water will evaporate into the air over hours/days. I am wondering as to the rate at which this evaporation happens, as an approximate percentage of the rate at which the mini-split wall unit is typically condensing latent moisture/water from the same area.
Essentially if the rate at which the condensate in the sink adds to the latent load is high, we will stop capturing the condensate in the sink and simply let the condensate drain thru the kitchen drain by removing the stopper.
Thanks.
Got it, Venkat.
In other words, you are trying to make sure that by saving the condensate to wash the dishes, you are not making the minisplit run more.
Under the right conditions, standing water can add a lot of humidity to an indoor space. That's one of the reasons why standing water in a basement can cause such damage even when it is not in direct contact with finish materials.
I don't know how to do the calculations, but I assume that if another GBA member does, they'll probably need to know some more details, like the temperature in the room, the relative humidity, the temperature of the water, etc.
I'm curious to see what others have to say about this.
I'd be more concerned that there could be nasty stuff growing in that condensate water. It's quite possible to have something like Legionella bacteria growing there. If it were me, I'd not use that water in any potable or semi-potable use. I'd use that water to water my house plants and leave it at that.
An easy way to check would be to fill a container with a known quantity of water, let it sit, and see how long it takes to evaporate.
I'd personally measure using a scale because it would be easier to measure small changes. (1g=1ml=1cc h2o).
If you really wanted to geek out, plot the conditions in the room, translate the change in evaporation into grains of h20 and see what change in RH results in.
If you want to know how much evaporate, I suggest you measure how much water evaporates from a container of water with the same surfaces area as your sink, mostly it depends on the temp and humidity in your home.
From a micro energy point of view if you hold the cold condensate water until it reaches room temp you could extract 1 BTU for each degree each pound of water is below the air temp. So if you collected a gallon of water at 60° in a room at 79° = 152 BTUs.
I think saving the water to clean your dishes is not a good idea as all the pollutant in your air will be concentrated in that water. The fan blows air across the wet coils anything in the air will stick to the water and be trapped in the water.
If you wanted to water your plants with the water I say go for it but do not drink the water or eat from surfaces contaminated with that water.
From most water supplies your cost for an additional gallon will be less than a penny each. Seems like a lot of work for little return.
Walta
"mostly it depends on the temperature and humidity in your home"
The evaporation rate depends strongly on the local wind speed at the surface of the water, which is challenging to measure. Also RH, but not so much temperature within normal indoor temp range. I strongly agree that the value can be determined very simply by the experiment you suggest. Also agree that the water should not be used, for anything really. I once met a hearty Vermont woman who collected her dehumidifier condensate for her laundry, but I don't see the point.
Condensate is nasty stuff. I would NOT use it to wash dishes. Condensate is corrosive, tends to have stuff growing in it (often microscopic critters that you can't see), and tends to have random things dissolved in it that came from the evaporator coils. You really don't want to use condensate to do anything that you want to keep clean. Best to just let it run down the drain.
Bill
Thanks all for the info and comments. Although after preliminary brush-cleaning of the dishes with the condensate water, the dishes are run thru the dishwasher, because of the cautions about Legionella, etc., that have been brought up, we will stop messing with the condensate. Thanks again.
I don't think the legionella concern is legitimate. The condensate comes off cold, and legionella is dormant at 20 C or below. It will grow a little above 25 C, but does't grow much until you are above 30 C.
As far as avoiding evaporation, I'd direct it into a jug with a smallish (~1" diameter) neck. That will drastically reduce evaporation, which would be slow anyway since it comes off cold.