Image Credit: Rocky Mountain Institute
Image Credit: Rocky Mountain Institute A chart from FDR Design showing the impact of gas fills and the spacing between the layers of glass in an IGU.
Image Credit: FDR Design, Inc.
Over the last two weeks I’ve covered the major strategies for improving the energy performance of windows: adding extra layers of glass, increasing the thickness of the air space between the layers of glass, and adding low-emissivity coatings. Another important strategy is to use a low-conductivity gas instead of air in the space between the layers of glass. Most commonly argon is used, though krypton is available for the highest-performance windows, and xenon is occasionally used.
Low-conductivity gas-fills don’t make as much difference as adding an additional layer of glazing, increasing the spacing between the layers of glass from a quarter- to a half-inch, or adding a low-e coating, but they are nonetheless significant — and definitely worth it when choosing new windows that have low-e coatings. Adding argon is the most cost-effective improvement you can make to a window. But what are these gas fills, and how do they work?
Why low-conductivity gases make sense
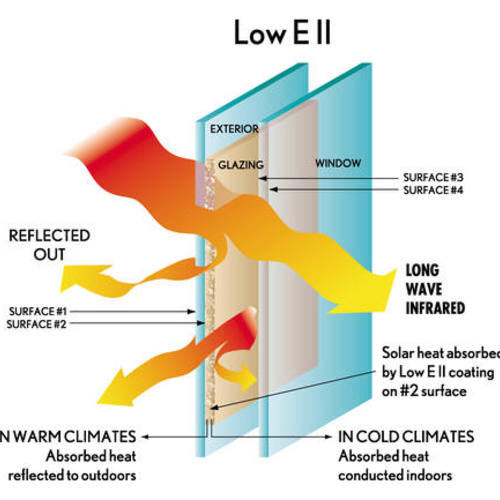
To understand how argon works, we have to go back to how heat moves through windows. There are three modes of heat transfer: conduction, convection, and radiation. With clear (non-low-e) double-glazed windows, radiation accounts for about half of the heat transfer, with conduction and convection each accounting for about 25%.
When a low-e coating is added to a window (see last week’s blog), the radiant component of that heat loss is significantly reduced, and as a result the conductive and convective portions become much more significant. As the name implies, low-conductivity gas fills reduce conductive heat flow. Most of us think of conduction, which is the transfer of kinetic energy from molecule to molecule, as occurring only through solids — think of a hot cast-iron skillet handle — but conduction also occurs across gases. Sometimes we refer to this as gas-phase conductivity.
The noble gases
Air has a thermal conductivity of 0.014 Btus per square foot per hour for every degree Fahrenheit difference in temperature (don’t worry about those units) at room temperatures. If we can replace that air with a lower-conductivity gas, we can slow the heat loss through windows. Argon is a great option. It has a conductivity of 0.0092 — 34% lower than that of air — and it is, by far, the most common low-conductivity gas for windows.
Some of the highest-performing windows use the more exotic gas, krypton, in the space between the glass. Krypton has a conductivity of 0.0051, which is 63% lower than that of air. Xenon, an even rarer gas, has a conductivity that’s 79% lower. These gases — found in the far-right-hand column of the Periodic Table — are all highly stable and unreactive, an attribute that earned them the moniker of “noble gases” (so named because, like nobility, they don’t interact with commoners).
All of these gases are components of the air we breathe. Argon makes up a little less than one percent of our atmosphere (third after nitrogen and oxygen) and is produced quite inexpensively as a byproduct of extracting oxygen out of the air. Krypton is present in air at a concentration of about one part per million (one ten-thousandths of a percent), and xenon is present at an even smaller concentration. As a result, these exotic gases are far more expensive to extract.
Buying a window filled with krypton instead of argon adds about $100 to the price, according to a Marvin Windows and Doors rep I spoke with recently, while there is little if any additional cost for argon. At a manufacturing cost of only about 10¢ per window, it’s one of the best deals around, according to Randi Ernst, president of FRD Design, Inc., which sells gas-filling equipment to the window industry.
The benefit of low-conductivity gases
Adding argon to a double-glazed window reduces the U-factor by about 0.05 (reducing the U-factor means reducing heat flow). With non-low-e glass, adding argon drops the U-factor from 0.50 to 0.45, a 10% reduction in heat loss (assuming optimal spacing for the glass).
When there’s a low-e coating, that same argon improves the U-factor from 0.30 to 0.25 — a much more respectable 17% improvement in performance. Using krypton with an optimal spacing drops the U-factor by another 0.025, so the total improvement over air is 25%.
The optimal thickness for gas fill
With an insulating glass unit (IGU), there is an optimal thickness that varies according to the gas fill. With a thicker air space there’s less conductive heat loss, but if the spacing gets too deep convective loops form that begin increasing heat loss (see my blog two weeks ago). With air, the optimal thickness for the air space is about a half-inch — assuming the standardized temperature conditions used for modeling window performance in this country. Argon is about the same — just a few millimeters thinner.
Significantly, if we assumed a lower difference in temperature (delta-T) between the indoors and outdoors, as they assume in Europe, there would be less convection between the glass and the optimal thickness would be greater — as we find on European windows. Because most of the U.S. actually experiences a significantly lower delta-T than the 70°F assumed in U.S. standards, a thicker glazing spacing actually makes sense.
With krypton, though, the optimal thickness is significantly less: about 5/16th of an inch (with U.S. delta-T assumptions). This is because krypton is more slippery than air or argon. It forms convective loops more easily, which increases that convective component of heat flow.
Do we really want radioactive windows?
It is a relatively little-known fact that krypton is somewhat radioactive. There are a lot of isotopes of krypton; krypton-85 with a half-life of 10.8 years, is the one that raises concern. Krypton-85 is produced by the fission of uranium and plutonium, and it gets released in the atmosphere through nuclear bomb testing, releases by nuclear power plants, and by the reprocessing of spent nuclear fuel.
The latter source is the most significant, and a majority of that comes from the French reprocessing plant, Cogema La Hague, which has been operating since 1976. The concentration of krypton-85 in the atmosphere has increased several-hundred-fold since the early 1940s, and some of that krypton-85 ends up in the krypton we extract from the atmosphere. As a result, canisters of krypton gas have measurable levels of radioactivity.
Is this significant for us, though? Probably not very. In most areas, the radioactivity from krypton in our windows will be lower than background radiation. If we’re willing to live with other sources of radiation in buildings, such as concrete foundations and granite countertops, we probably shouldn’t worry too much about krypton. However, ionizing radiation is cumulative, and when we can avoid exposure we should try to do so.
Does the gas stick around?
The question of whether the low-conductivity gas lasts in an IGU is huge. If it leaks out in a few years, it wouldn’t be worth spending more for it. The rule-of-thumb, based on laboratory testing, is that 1% of the gas will be lost per year. Oddly, there has been very little research done on gas retention rates in the field.
Fortunately, the research that has been done offers generally good news. Randi Ernst has done about the only field testing of gas retention rates that I know of. From repeatedly testing several dozen windows over a period of years, he has found that about 0.6% of the gas leaks out per year.
That’s a pretty low leakage rate: a window starting with 95% argon would be down to 79% argon after 30 years and 70% argon after 50 years. Even assuming 1% annual loss, after 30 years, there will still be 70% of the original argon, and after 50 years 58%. Most windows don’t last 50 years for other reasons, so I’m comfortable with the gas retention.
The bottom line
It’s always worth adding low-conductivity gas fill to an IGU.
While I’m not terribly worried about the radioactivity of krypton, it does give me pause, and we get far more bang for the buck with argon. If I want better energy performance than can be achieved with low-e and argon in an IGU, rather than replace the argon with krypton, I’ll specify a third layer of glass with another low-e coating or a second low-e coating on the inner (#4 surface) of a double-glazed IGU (see last week’s blog).
Alex is founder of BuildingGreen, Inc. and executive editor of Environmental Building News. To keep up with his latest articles and musings, you can sign up for his Twitter feed.
Weekly Newsletter
Get building science and energy efficiency advice, plus special offers, in your inbox.














10 Comments
More about radioactive Kr-85
Nice article, Alex. On the radioactivity of Kr-85, it's a β− emitter, and the β− particles (electrons) are of relatively low energy (251 keV average, 687 keV max). As long as the gas is enclosed in the IGU, the radiation is most likely all absorbed by the glass and window frame and never gets a chance to ionize the atoms in anyone's body. You could hold a Geiger counter up next to the window, and it would probably not register a single count due to the Kr-85 inside the IGU.
With the low leakage rate you cited above, only a little bit of Krypton will get into the air in the house, and that little bit contains only a small amount of Kr-85. Once in the house, it has a greater-than-zero chance of damaging cells -- but it's a chance that's barely above zero because of the tiny, tiny amount mixed in with all the house air. That's another reason for a good ventilation system, but even without it, the chances of Kr-85 causing harm are slim to none.
So, you're right not to worry about the radioactivity of Kr-85. I doubt I'd use windows filled with Krypton, though, just because of the extra expense.
Optimal Spacing
To paraphrase Osbourn's Law,
It's amazing how many times variables won't, and constants aren't.
Calculating Uwindow requires an assumption about the indoor-outdoor temperature difference. Historically Uwindow was used to size heating equipment, so quite logically the the indoor-outdoor temperature difference was based on a coldest day scenario.
But as Alex points out:
“Significantly, if we assumed a lower difference in temperature (delta-T) between the indoors and outdoors, as they assume in Europe, there would be less convection between the glass and the optimal thickness would be greater — as we find on European windows. Because most of the U.S. actually experiences a significantly lower delta-T than the 70°F assumed in U.S. standards, a thicker glazing spacing actually makes sense.”
.
Which to me prompts the question – what is the more pressing need?
.
An accurate Uwindow to prevent undersized heating equipment?
(which means using a worst case delta-T)
- or-
An accurate Uwindow to predict annual energy consumption?
(which means using an average case delta-T)
.
If the answer is the latter rather than the former, then the NFRC should be adopting the CEN approach of basing U-window on a delta-T of 36F, instead its current delta-T of 70F.
.
.
Stephen Thwaites
Thermotech Fiberglass Fenestration
.
P.S. Here in Ottawa, Ontario (the 3rd coldest national capital and home to the world's largest outdoor skating rink), the average heating season temperature delta-T is only 39F.
re
A link to the whole set of graphs like the one above[with some other editorial content]:
http://www.igasfill.com/images/stories/gas_filling_of_ig_units_-_full_web_version.pdf
would be pretty interesting to understand the whole 3 dimensional graph of delta T glass spacing, spacer types etc.
more stuff:
http://www.nfrc.org/documents/IGMASpacerStudyfinalreport.pdf
Thanks Stephen!
You've nicely answered the question I posted on the first article in this series on why the Germans and Austrians build thicker IGU's. Muchas gracias from a sunny NorCal where our delta T over the past few chilly days has been around 23F: daytime temps in the low 60's and nights hovering around 40F. Yeah, poor us, but still worthy of better windows!
detailed info glazing spacing etc
Have not finished reading it, but interesting. Cannot see a date, but looks older, references from late 80's
http://www.google.com/url?sa=t&rct=j&q=graph%20%22u%20value%22%20glass%20spacing&source=web&cd=1&ved=0CCAQFjAA&url=http%3A%2F%2Fsolarme.uwaterloo.ca%2FDownloadPDFs%2FPaneSpacing_SESCI.PDF&ei=PtqCT9mxNomy0QG2-IDBCQ&usg=AFQjCNGhdNikDipC9Ay3c-WHI7k8uqkN4A&cad=rja
This and that...
Alex,
Those were three really great blogs! The second one on coatings was as good as I remember ever having read online before. Thanks!
A couple random thoughts...
There are of course six named noble gasses listed in the periodic table - and currently argon, krypton, and xenon are available as IGU fills if some one wants to spend the money. Imagine the next noble gas on the list as an IGU fill...imagine being the marketing director and trying to sell that one to people?
You wrote a great blog on vacuum glazing a while back, and now the three-parter, so I am curious if you have had any thoughts about writing anything about some of the other, more exotic, glazing options out there? Thermochromic, electrochromic, raidant heated glass, are just three that immediately come to mind...
Andersen's triple-glazed window
David,
Thanks for your comments. I learned a few weeks ago that Andersen is coming out with a new triple-glazed window (finally) this fall. With codes getting tighter, they are having to pay attention and get on board. I'll look forward to seeing it.
BTW, I'm working on an article on passive-house-appropriate windows and would be interested in your experiences with different products. Feel free to e-mail me with any thoughts you might have: [email protected]. (Anyone else too!)
Optimal Spacing
Stephan has some excellent points about the influence of boundary conditions. I would say that convection, not conduction, is always driving the heat transfer between the panes at center glass. Even at the lower temperatures, ignoring the convection will lead to error. However, the natural convective effect gets greater as the temperature differences increases. Also, the improvement in performance with Krypton is reduced as the temperature differences increase.
I agree with the assessment about calculations for peak loads versus average loads. However, I think the former is more important. Maybe we can get the NFRC to require graphs of U-Factor versus temperature difference :)
David Paulus
WASCO Windows
Very informative blog. I
Very informative blog. I have recenty installed suspended film passive house standard windows and will install the European triple glaze windows on the another project in the coming months. The Europeans seem to be very skeptical about the longevity of those films.
I enjoyed the technical arguments in the blog but i can't help but remind everyone hear that the larger market for windows can never be penetrated by a window however good the glazing, if it costs a small fortune and the addditional parts needed cost another fortune oh and don't forget the fork lift!!! ever pick up a triple glazed 6''x5' picture window made in Europe? Uggg!! my back, my neck, my leg!!!
Noboby wants a window that can't withstand the element no matter how good the glazing! Agreed?
Please Anderson get involved already!! Build the window everyone on this site knows you could build and make them afordable at the same time, its really not that hard i've held all the great designs in my hands! ........well.....we're waiting......forgive my enthusiasim but this is very troubling subject for me. thanks for reading..
Glazing efficiency
The windows being built today while more efficient are still holes in walls that heat pours through! It seems to me that with the technology available today regarding "heat transfer" can be used in creating windows that can capture the heat that rises to the top of the window and using the same techniques as a heat pump or refrigeration unit to move the heat back into the structure. Micro refrigeration units are available today and could make windows as well as exterior walls into heat recovery systems.
Log in or create an account to post a comment.
Sign up Log in